 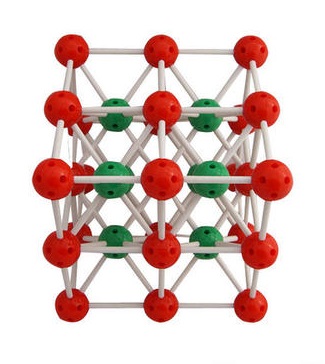
The HCP crystalline structure has few ways that slipping can occur, giving these materials a high strength but typically a brittle failure mode. Cobalt, zinc and zirconium are a few other well-known examples. Titanium and cadmium are two of the most commonly used metals that are comprised of the HCP crystalline structure at room temperature. The HCP crystalline structure is found in several different metals. The atoms in the hexagons are shared with adjacent HCP structures. Sandwiched, or packed, in between these two hexagons is a group of three atoms that are not in line with the atoms in either of the hexagons. Cesium chloride CsCl or ClCs CID 24293 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities. From this figure, is one-half of the bond angle or 109.5 /2 54.75, which means that 90 54.75 35.25 since the triangle shown is a right triangle. Both groups of atoms in the hexagons are aligned with one another. The drawing below shows the cubic unit cell with those carbon atoms that bond to one another in one-quarter of the unit cell. Additionally, there is an atom in the center of each of these hexagons. An additional six atoms are distributed equally across the vertices of another hexagon. There are six atoms spread evenly among each vertex of a hexagon. If one hexagonal close-packed structure were separated from other hexagonal close-packed structures adjacent to it, it would be comprised of 17 atoms. Hexagonal close-packed (HCP) is a crystalline structure that is somewhat more complex than the FCC and BCC crystalline structures. This atom is centered in the body of the cube, which is the reason for the name body-centered cubic. What is different about the BCC crystalline structure is that rather than having an atom at each of the six faces, it has only one atom that is inside the cube. The BCC crystalline structure is in the form of a cube with eight atoms distributed among the eight corners similar to the FCC crystalline structure. Like the FCC crystalline structure, the BCC crystalline structure gets its name from its shape. The body-centered cubic (BCC) crystalline structure is another abundant type of atomic structure found in metals. (Learn more in the article An Introduction to Stainless Steels.) Body-centered Cubic An example of this is austenitic stainless steel. Adding certain alloying elements (e.g., nickel) to steel allows for steel to be austenitic, and therefore FCC, at room temperature. Iron does not have the FCC crystalline structure at room temperature, but when heated to a certain temperature, the typical ferrite body-centered cubic found in iron begins to transform to austenite, which does have an FCC crystalline structure. Nickel and precious metals such as gold, platinum and silver have the FCC crystalline structure as well. The two examples in the introduction, aluminum and lead, are two metals that have the FCC structure at room temperature. There are many different types of metal with the FCC crystalline structure. In addition to the atoms located on the corners of the FCC structure, there are also six atoms located on the center of each cube face, hence the name face-centered cubic. Each of those eight atoms are part of other adjacent cubic structures as well. There are eight atoms that are distributed among the eight corners of the crystalline structure. The FCC crystalline structure gets its name from its cube shape and the locations of the atoms within that cube. One of the most common crystalline structures is face-centered cubic (FCC). The three most common crystalline structures in metal are face-centered cubic, body-centered cubic and hexagonal close-packed. Within the crystalline structure group there are a range of subgroups that organize the shape of these crystalline structures. This means that the atoms of metals are arranged in a patterned, three-dimensional way that repeats itself throughout large portions of the metal. However, metals in particular almost always have their atoms organized in a crystalline lattice structure. There is one atom in CsCl.Materials are made up of a wide variety of atomic structures. The cations are located at the center of the anions cube and the anions are located at the center of the cations cube. Each Cs + is sur rounded by 8 Cl - at the corners of its cube and each Cl - is also surrounded by 8 Cs + at the corners of its cube. \): Unit Cell of CsCl (en./wiki/Caesium_chlorideĪnions and catio ns have similar sizes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
